The Science Behind Our Tests:
Empowering Your Fertility Journey
Powered by SpermComet® Technology
Examen’s sperm DNA tests, powered by our proprietary SpermComet® technology, are trusted by fertility experts in 80% of the UK & Ireland’s top private fertility clinics. We provide highly accurate and sensitive tests to guide your next steps toward parenthood.
The Role of DNA in Sperm Cells
Every cell in our bodies, except for red blood cells, contains DNA in its nucleus. This DNA holds all the information needed for each cell to function. To fit into the nucleus, DNA is carefully packed onto chromosomes (23 pairs in total). However, this packing must strike a balance – tight enough to protect the DNA but loose enough to access the genetic information when needed.
Sperm cells are unique. Their sole purpose is to deliver 50% of the baby’s genetic blueprint (one set of 23 chromosomes) from the father to the egg, which contains the other 50% from the mother. To ensure the DNA reaches the egg safely, it is tightly packed for the journey – akin to walking from Land’s End to John O’Groats and back four times! Like a Formula 1 car, sperm cells strip away everything unnecessary for the journey, including the machinery that repairs DNA damage.
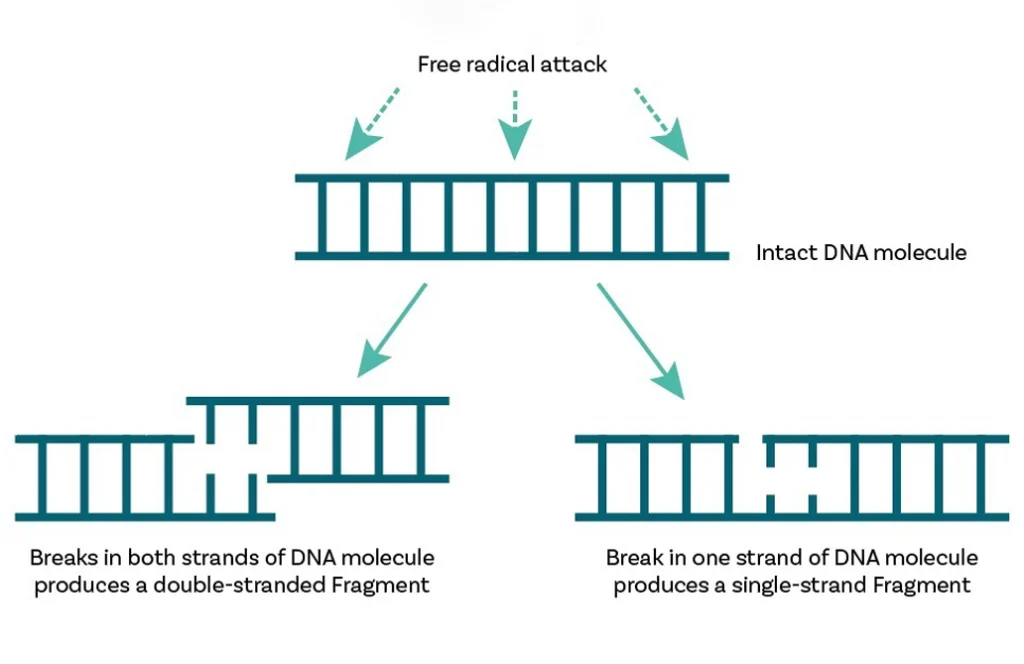
How DNA Damage Affects Sperm
DNA exists as two strands arranged in a double helix, resembling a spiral staircase. Damage to this structure can occur in two ways:
- Single-strand breaks (ssDB): More common and less impactful.
- Double-strand breaks (dsDB): Less common but more challenging for the sperm to overcome.
Sperm cells are particularly vulnerable to DNA damage because they lack the repair mechanisms found in other cells. This vulnerability is due to the dense packing of DNA and the absence of cellular machinery that prevents or repairs damage. Factors such as lifestyle choices, stress, and medical conditions can exacerbate this damage.
The Science Behind SpermComet® Technology
Examen’s tests are powered by SpermComet® technology, which is based on a well-established scientific technique called single cell gel electrophoresis. This method allows us to measure DNA damage in individual sperm cells, providing unparalleled accuracy and insight.
How SpermComet® Works
1
Relaxing the DNA Structure
A semen sample is gently processed to relax the dense DNA structure in each sperm cell.
2
Electrophoresis
The DNA is exposed to an electric field, causing damaged fragments to migrate out of the sperm head.
3
Comet Formation
Under a microscope, damaged DNA appears as a visible “comet” shape, with the head representing intact DNA and the tail representing fragmented DNA.
This detailed analysis of individual sperm cells ensures the most accurate results, setting Examen apart from other tests that rely on bulk measurements or automated processing.
Measuring Total and Double-Strand DNA Damage
Our tests are particularly valuable for understanding the underlying causes of fertility challenges and tailoring treatment plans.
Measures total DNA damage, including both single- and double-strand breaks.
Specifically measures double-strand break damage, providing deeper insights into sperm DNA integrity.
Why Human Expertise Matters
Unlike automated methods, the SpermComet® technique incorporates a high degree of human skill and experience. Our laboratory experts analyse each sample with precision, ensuring reliable results. This human expertise is what makes Examen’s tests the most accurate male fertility tests available.
Why Sperm DNA Quality Matters
Traditional semen analysis focuses on parameters like sperm count, motility, and morphology, but it does not assess DNA quality. Even if a semen analysis result is “normal”, there could still be issues with sperm DNA integrity that impact fertility. This is why Examen’s SDF tests are so important – they provide a deeper understanding of sperm health.

Medical Research
- Osadchuk L, Kleshchev M, Osadchuk A. Effects of cigarette smoking on semen quality, reproductive hormone levels, metabolic profile, zinc and sperm DNA fragmentation in men: results from a population-based study. Front Endocrinol (Lausanne). 2023 Oct 18;14:1255304. doi: 10.3389/fendo.2023.1255304. PMID: 37920251; PMCID: PMC10619690. ↩︎
- Çayan S, Farkouh A, Agarwal A, Atmoko W, Wyns C, Arafa M, Zini A, Shah R, Alipour H, Chung E, Saleh R, Pinggera GM, Konstantinidis C, Al Hashimi M, Pescatori E, Rambhatla A, Toprak T, Calogero AE, Gül M, Park HJ, Altay B, Falcone M, Rashed A, Le TV, Bahar F, Shatylko T, Görür S, El-Sakka AI, Saylam B, Sarikaya S, Smith RP, Boeri L, Efesoy O, Ceyhan E, Russo GI, Ozer C, Ho CC, Gungor ND, Özlü DN, Molina JM, Musa MU, Tsujimura A, Gokalp F, Mohamed MS, Okada K, Khalafalla K, Kuroda S, Binsaleh S, Motawi AT, Shamohammadi I, Mogharabian N, Manh MT, Taha EA, Makarounis K, Mak SK, Shedeed SA, Thomas C, Mostafa T. Global Andrology Forum Clinical Guidelines on the Relevance of Sperm DNA Fragmentation in Reproductive Medicine. World J Mens Health. 2025 Apr;43:e21. https://doi.org/10.5534/wjmh.250005 ↩︎
A Commitment to Scientific Excellence
Examen is committed to advancing the field of male fertility diagnostics through rigorous scientific research and innovation. Our ISO-accredited laboratory and proprietary SpermComet® technology ensure that every test we perform meets the highest standards of accuracy and reliability.
Test with scientific confidence
Don’t let sperm DNA damage stand in the way of your dreams. Book your SDF test with Examen and gain the clarity you need to move forward.
Fast, reliable, and ISO-accredited testing.